Inhalation toxicity of surfactants
Abstract
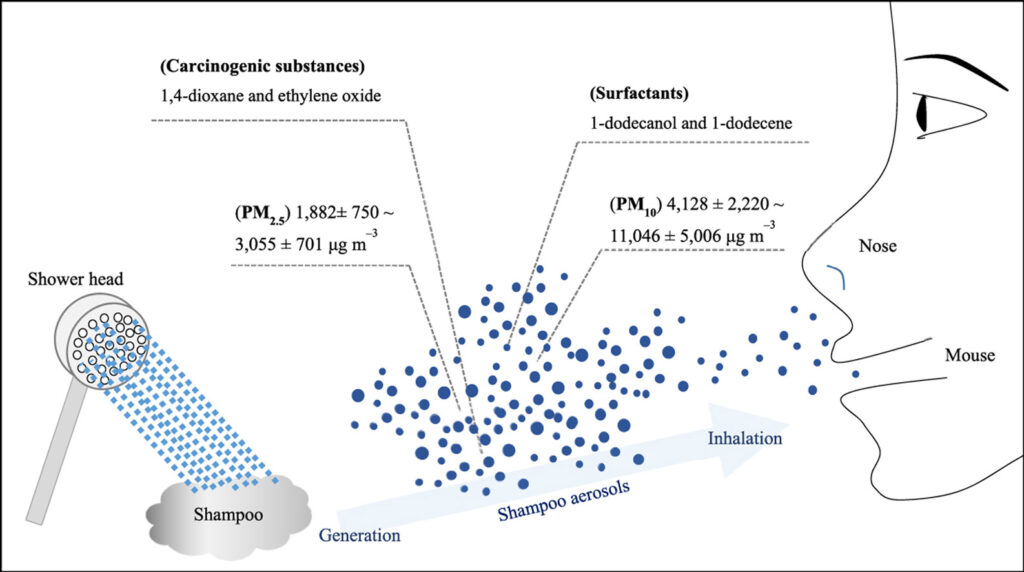
To declare a shampoo toxicologically safe, one should evaluate the hazards posed by the inhalation of aerosols produced during its use. Herein, tap water was sprayed into a shampoo-filled plastic container to investigate the formation of shampoo aerosols and the possibility of their inhalation. The aerosols thus obtained had higher mass concentrations (geometric mean = 5779 μg m−3 (PM10) and 2249 μg m−3 (PM2.5)) than water aerosols (geometric mean = 927 μg m−3 (PM10) and 476 μg m−3 (PM2.5)). In particular, shampoo aerosol particles with an aerodynamic diameter of 2.5 μm, which can penetrate the alveoli when inhaled, had the highest mass concentration (geometric mean = 2000 μg m−3). The volatile organic compounds contained in shampoo aerosols featured alcohol and ether groups attached to dodecane and tetradecane backbones; these compounds were generated by the thermal decomposition of surfactants (i.e., lauryl and laureth sulfates) during instrumental analysis. The acquired data suggest that inhalation exposure and chronic inhalation toxicity evaluations should be performed for various shampoo usage conditions to ensure inhalation safety.
Introduction
In view of the widespread use of household chemical products in living environments, the hazards posed by the chronic exposure to these products are being explored (Bearth et al., 2020; Gummin et al., 2017; Kim et al., 2013; Lee and You, 2020) and they justify the strict enforcement of related regulations. However, the efficient compliance with such regulations and their establishment requires the identification of the harmful components of these products and the detailed evaluation of the related exposure hazards. Among other reasons, the fact that the toxicities of these components significantly depend on the exposure route, i.e., inhalation, ingestion, or dermal contact (Gagnon and Fromm, 2015; Galić et al., 2020; Joshi and Adhikari, 2019; Krishtal et al., 2019; Zhang et al., 2010), complicates the said evaluation.
The harmfulness of shampoo, a representative household chemical product, has been extensively studied, as exemplified by the numerous works involving the analysis of its major components and harmful ingredients, toxicity testing, and safety assessment (Guzmán and Santini, 2019; Hidalgo et al., 2017; T Chiu et al., 2015). Surfactants, ethylene oxide, and 1,4-dioxane have been identified as the main harmful components of shampoo, with surfactants having the largest mass content (Jinot et al., 2018; Li et al., 2018; Mainkar and Jolly, 2001; Zenker et al., 2003), which mainly consist by sodium lauryl sulfate (SLS), ammonium lauryl sulfate, sodium laureth sulfate (SLES), and ammonium laureth sulfate (Jurek et al., 2019). According to the Globally Harmonized System, these four surfactants have low or no acute toxicity via inhalation, ingestion, and dermal exposure (Kim et al., 2013; Petit et al., 2017). However, in view of the chronic exposure of most humans to shampoo, repeated toxicity studies are needed for these surfactants. In the case of oral exposure, the no-observed-adverse-effect level of SLS is 100–400 mg kg−1 day−1, which, in the case of shampoo, is difficult to achieve in practice, with notable exceptions being sub-chronic oral toxicity studies in rats using repeated doses (Kumar et al., 2014). However, in the case of inhalation exposure, data pertaining to the sub-chronic and chronic toxicity of surfactants are scarce, as it is assumed that the repeated inhalation exposure of humans to shampoo does not occur. Therefore, one needs to experimentally check whether inhalation exposure to shampoo is possible and thus confirm or disprove the necessity of acquiring repeated inhalation toxicity data for surfactants.
Herein, the possibility of inhalation exposure to shampoo was evaluated by simulating the actual use of shampoo, and the potential of exposure to the generated shampoo aerosol was evaluated by analyzing the aerosol particle size and composition. The obtained results demonstrate the need to inform consumers of the necessity of evaluating the risks posed by shampoo inhalation, and they can be used for the related risk assessment.
Experimental setup
Initially, we simulated the actual use of shampoo to confirm or disprove the generation of shampoo aerosols (Exp stage 1). When shampoo aerosols were generated, we conducted a comparative analysis to determine whether they contained shampoo components (Exp stage 2) (Table 1A).
In Exp stage 1, aerosol generation was induced by spraying tap water into a container either containing or not containing shampoo, and the number concentrations and sizes of aerosol particles were measured using an optical
Correlation of particle concentration with particle size
The number concentrations of water and shampoo aerosol particles were measured for different particle sizes and converted to mass concentrations using the following equation:
mass concentration (μg m−3) = 4/3πr3 (μm3 number−1) × density (g cm−3) × number concentration (number L−1) × (cm3/1012 μm3) × (103 L/m3) × (g/106 μg).
The aerosol particles were assumed to have a certain aerodynamic diameter, and their concentration was calculated using the minimum and maximum diameters of the OPC particle
Conclusions
This study probed the possibility of shampoo aerosolization and emphasized the need for chronic inhalation toxicity testing. Although the possibility of shampoo inhalation during use is not negligible, there are no data on the inhalation toxicity of shampoo; this inspired us to evaluate shampoo aerosol formation and inhalation potential. After the generation of a shampoo aerosol in a simulated shampoo use environment (water was sprayed into a shampoo-filled plastic bath), the aerosol particle